- Research Article
- Open access
- Published:
The Relationship between Changes in Weight Status and Insulin Resistance in Youth
International Journal of Pediatric Endocrinology volume 2009, Article number: 862061 (2009)
Abstract
This study examined changes in insulin resistance (IR) in 120 youth over two years. IR was quantified via homeostatic model (HOMA-IR), and weight status changes were quantified via body mass index (BMI). When all participants were considered, the mean HOMA-IR and BMI increased 13.4% and 1.65 units, respectively. Change in BMI z-score and percent change in HOMA-IR were moderately associated ( ). Follow-up analyses were performed for the following weight groups: NN (normal at baseline and two years later), NO (normal to overweight), ON (overweight to normal), and OO (overweight at both points). The NO group had a greater change in HOMA-IR (+50%) compared to other groups: ON (
). Follow-up analyses were performed for the following weight groups: NN (normal at baseline and two years later), NO (normal to overweight), ON (overweight to normal), and OO (overweight at both points). The NO group had a greater change in HOMA-IR (+50%) compared to other groups: ON ( 8%), NN (+2%), and OO (
8%), NN (+2%), and OO ( 0.1%) (
0.1%) ( ). The association between changes in BMI z-score and HOMA-IR was
). The association between changes in BMI z-score and HOMA-IR was  when only the NO and ON groups were included. These results reinforce the importance of preventing youth from becoming overweight to control IR.
when only the NO and ON groups were included. These results reinforce the importance of preventing youth from becoming overweight to control IR.
1. Introduction
Insulin resistance (IR) is a precursor to type II diabetes, and recent reports show that 52% to 72% of overweight and obese youth have IR [1, 2]. While numerous cross-sectional studies have noted positive associations ( to 0.85) between insulin and measures of adiposity in youth, the relationships between change in weight status and change in IR have not been examined thoroughly [2–8]. The few longitudinal studies of youth have consistently reported that change in weight is positively associated with change in insulin or IR and negatively associated with insulin sensitivity [9–11]. However, these studies have induced weight loss by an intervention and have not examined what occurs in response to natural changes in weight status.
to 0.85) between insulin and measures of adiposity in youth, the relationships between change in weight status and change in IR have not been examined thoroughly [2–8]. The few longitudinal studies of youth have consistently reported that change in weight is positively associated with change in insulin or IR and negatively associated with insulin sensitivity [9–11]. However, these studies have induced weight loss by an intervention and have not examined what occurs in response to natural changes in weight status.
The current investigation will add to our understanding of the relationship between changes in weight status and IR by directly comparing changes in IR in youth who increased, decreased, or maintained their weight status over a two-year period. Analyses were first conducted with all participants in one group. Secondary analyses were conducted to compare IR among four weight groups, as described in the Methods section. We hypothesized that a larger increase in IR would occur in the participants who were normal weight and became overweight compared to those who remained normal weight. Also, we expected that a larger decline in IR would occur in participants who were overweight and became normal weight compared to those who remained overweight two years later.
2. Methods
2.1. Participants
The participants were enrolled in the Cardiovascular Health in Children III (CHIC) study, Cohort 5 (J.S. Harrell, P.I.). The CHIC III study was a longitudinal investigation assessing risk factors for cardiovascular disease in youth from rural North Carolina, taking place from 2000 to 2003. Of the 1566 participants in Cohort 5, the mean ages at baseline and followup were  years and
years and  years, respectively. The sex and racial composition of this cohort was 50% female, 50% male, 53% Black, 36% White, and 11% Other races. A total of 120 children and adolescents were selected from the 1566 participants to participate in this study, based on their weight status as described in the Group Analyses section. The age of participants in this investigation was similar to the original cohort (mean ages
years, respectively. The sex and racial composition of this cohort was 50% female, 50% male, 53% Black, 36% White, and 11% Other races. A total of 120 children and adolescents were selected from the 1566 participants to participate in this study, based on their weight status as described in the Group Analyses section. The age of participants in this investigation was similar to the original cohort (mean ages  years at baseline and
years at baseline and  years at followup). There were slightly more males (62%), and White participants (56%), compared to the original cohort.
years at followup). There were slightly more males (62%), and White participants (56%), compared to the original cohort.
2.2. Procedures
All data were collected at baseline and two years later, termed baseline and followup in this study. Prior to data collection, the participants and their parents signed assent and consent forms, respectively, and all procedures were reviewed and approved by the Institutional Review Board at the University of North Carolina at Chapel Hill.
Data were collected at the participants' schools by trained research assistants. Height was measured using a stadiometer (Perspective Enterprises, Portage, MI), and body mass was assessed via electronic scale (Model 2101KL, Healthometer Medical, Bridgewater, IL). BMI was calculated from these measurements (kg/ ). Fat mass and fat free mass were estimated from measures of percent body fat, obtained from sum of skinfolds taken at the subscapular and triceps sites. These measures were taken in triplicate using Lange calipers (Cambridge Scientific, Cambridge, MD), according to the guidelines set forth by NHANES III [12]. Equations specific to each sex, race, and pubertal status were then applied to estimate percent body fat [13]. In order to ensure strong intra- and interrater reliability, respectively, all research assistants were trained by the same investigator, and height, body mass, and skinfolds were measured by more than one research assistant for every tenth participant. If the measurement for any variable was greater than pre-established quality control criteria, the research assistant was not permitted to participate in future measurements for that variable.
). Fat mass and fat free mass were estimated from measures of percent body fat, obtained from sum of skinfolds taken at the subscapular and triceps sites. These measures were taken in triplicate using Lange calipers (Cambridge Scientific, Cambridge, MD), according to the guidelines set forth by NHANES III [12]. Equations specific to each sex, race, and pubertal status were then applied to estimate percent body fat [13]. In order to ensure strong intra- and interrater reliability, respectively, all research assistants were trained by the same investigator, and height, body mass, and skinfolds were measured by more than one research assistant for every tenth participant. If the measurement for any variable was greater than pre-established quality control criteria, the research assistant was not permitted to participate in future measurements for that variable.
Researchers have noted that pubertal status [5, 7, 14, 15] and aerobic fitness [16] influence HOMA-IR; thus, both measures were included in this investigation. Pubertal development was quantified using self-report questionnaires [17]. Aerobic fitness was assessed using a multistage submaximal cycle ergometer test, the Physical Work Capacity 195 or  [18]. The workload corresponding to a heart rate of 150–170 bpm was used to predict the maximal volume of oxygen uptake (pV
[18]. The workload corresponding to a heart rate of 150–170 bpm was used to predict the maximal volume of oxygen uptake (pV max), and strong correlations (
max), and strong correlations ( ) have been shown between this test and measured V
) have been shown between this test and measured V max in children [18]. The results of the test were expressed in mL of oxygen per kg fat-free mass per minute (V
max in children [18]. The results of the test were expressed in mL of oxygen per kg fat-free mass per minute (V /
/ ), to eliminate the effect of fat mass.
), to eliminate the effect of fat mass.
Blood was drawn using standard venipuncture methods, between 7 and 9 am, following an 8-hour fast. The samples were centrifuged, and the plasma was stored in vials and frozen in a  freezer for later analysis. Insulin was measured from the stored plasma using radioimmunoassay procedures (Linco, St. Charles, MO), while glucose was analyzed via automated hexokinase oxidase procedures. These values were used to calculate IR using the homeostatic model assessment equation (HOMA-IR); (fasting insulin (
freezer for later analysis. Insulin was measured from the stored plasma using radioimmunoassay procedures (Linco, St. Charles, MO), while glucose was analyzed via automated hexokinase oxidase procedures. These values were used to calculate IR using the homeostatic model assessment equation (HOMA-IR); (fasting insulin ( U/mL) multiplied by fasting glucose concentration (mmol/L))/22.5 [19]. Several recent investigations have shown that HOMA is both reliable and valid for use in pediatric populations [20–22].
U/mL) multiplied by fasting glucose concentration (mmol/L))/22.5 [19]. Several recent investigations have shown that HOMA is both reliable and valid for use in pediatric populations [20–22].
2.3. Statistical Analysis
Initially, the statistical analyses were performed with all participants combined into one group ( ). The percent change in HOMA-IR ((follow-up − baseline)/baseline) was calculated for each participant. Likewise, change in weight status was analyzed by calculating the z-score of BMI change (follow-up BMI z-score minus baseline BMI z-score). The relationship between percent change in IR and change in BMI z-score was examined using Pearson correlations. This association was then examined after controlling for change in pubertal status and aerobic fitness, expressed in mL/
). The percent change in HOMA-IR ((follow-up − baseline)/baseline) was calculated for each participant. Likewise, change in weight status was analyzed by calculating the z-score of BMI change (follow-up BMI z-score minus baseline BMI z-score). The relationship between percent change in IR and change in BMI z-score was examined using Pearson correlations. This association was then examined after controlling for change in pubertal status and aerobic fitness, expressed in mL/ /min (V
/min (V /
/ ).
).
To establish which variable had the greatest influence on HOMA-IR, we repeated these correlation analyses using percent changes in insulin and glucose as the dependent variables. The alpha level was set at  , and 95% confidence intervals were computed for the correlations. All statistical analyses were conducted using SAS Statistical Software, Version 9.1 (Cary, NC).
, and 95% confidence intervals were computed for the correlations. All statistical analyses were conducted using SAS Statistical Software, Version 9.1 (Cary, NC).
2.4. Group Analyses of Weight Status Change
For the group analyses of weight status change, participants were divided into four groups (NO, NN, ON, OO) based upon their BMI percentile at baseline and followup. BMI percentile was derived from the Centers for Disease Control and Prevention (CDC) growth charts from the year 2000 [23]. Normal weight was defined as  5th and
5th and  85th BMI percentile for age and sex, while overweight was defined as
85th BMI percentile for age and sex, while overweight was defined as  85th BMI percentile for age and sex. The NO group was comprised of participants (
85th BMI percentile for age and sex. The NO group was comprised of participants ( ) who were normal weight at baseline and overweight at followup. These individuals were selected from participants in the original cohort who met these criteria (
) who were normal weight at baseline and overweight at followup. These individuals were selected from participants in the original cohort who met these criteria ( ) and had complete data (35 out of 60). A comparison group, NN, was comprised of 35 participants who were normal weight at both time points. This group was randomly selected from participants from the original cohort who matched those in the NO group by sex, race (Black, White, or Other), and pubertal status at baseline. Conversely, the ON group was comprised of 25 participants who were overweight at baseline and normal weight at followup and had complete data available (25 out of 35 total). Participants for the corresponding comparison group, OO, (
) and had complete data (35 out of 60). A comparison group, NN, was comprised of 35 participants who were normal weight at both time points. This group was randomly selected from participants from the original cohort who matched those in the NO group by sex, race (Black, White, or Other), and pubertal status at baseline. Conversely, the ON group was comprised of 25 participants who were overweight at baseline and normal weight at followup and had complete data available (25 out of 35 total). Participants for the corresponding comparison group, OO, ( ) were randomly selected from eligible participants who matched those in the ON groups by sex, race, and pubertal status at baseline.
) were randomly selected from eligible participants who matched those in the ON groups by sex, race, and pubertal status at baseline.
A one-way ANOVA was computed to compare the following variables between the four weight groups at baseline: age, pubertal stage, body mass, height, BMI, BMI percentile, V /
/ , glucose, insulin, and HOMA-IR. A Mantel-Haenszel chi-square was calculated to assess group differences at baseline for the categorical variables race and sex. To assess group differences in percent change in HOMA-IR and BMI change z-score, one-way ANOVAs were computed. Finally, the association between percent change in IR and change in BMI z-score was examined for the two groups that altered their weight status (NO and ON), using Pearson correlations. Partial correlations between these variables were calculated after controlling for changes in pubertal status and V
, glucose, insulin, and HOMA-IR. A Mantel-Haenszel chi-square was calculated to assess group differences at baseline for the categorical variables race and sex. To assess group differences in percent change in HOMA-IR and BMI change z-score, one-way ANOVAs were computed. Finally, the association between percent change in IR and change in BMI z-score was examined for the two groups that altered their weight status (NO and ON), using Pearson correlations. Partial correlations between these variables were calculated after controlling for changes in pubertal status and V /
/ . These correlations were repeated using percent change in insulin and glucose, respectively, in place of percent change in HOMA-IR to determine which variable was driving the change in HOMA-IR.
. These correlations were repeated using percent change in insulin and glucose, respectively, in place of percent change in HOMA-IR to determine which variable was driving the change in HOMA-IR.
3. Results
3.1. All Participants
Descriptive characteristics of all participants are presented in Table 1. With the exception of a fairly high BMI, none of the other characteristics were remarkable. The Pearson correlation between BMI change z-score and percent change in HOMA-IR was 0.39,  . After controlling for changes in pubertal status and aerobic fitness (V
. After controlling for changes in pubertal status and aerobic fitness (V /
/ ), the partial correlation increased slightly to 0.40;
), the partial correlation increased slightly to 0.40;  (Table 2).
(Table 2).
 /
/
The correlations between percent change in glucose and BMI change z-score were not significant when all participants were included ( ); therefore no further analyses were conducted using glucose. However, the Pearson correlations and partial correlations for BMI change z-score and percent change in insulin were significant and nearly identical to those for percent change in HOMA-IR (Table 2).
); therefore no further analyses were conducted using glucose. However, the Pearson correlations and partial correlations for BMI change z-score and percent change in insulin were significant and nearly identical to those for percent change in HOMA-IR (Table 2).
3.2. Weight Group Analysis
Descriptive characteristics of the four weight groups are presented in Table 3. When comparing the four groups, there were no differences for sex but there were significant differences for race, such that the OO and ON groups had fewer Black participants than the NN and NO groups ( :
:  ). Despite similar medians and ranges of pubertal stages in all four groups (
). Despite similar medians and ranges of pubertal stages in all four groups ( ), participants in the NO were approximately one year younger than participants in the other groups (
), participants in the NO were approximately one year younger than participants in the other groups ( ). There were no group differences in glucose at baseline but the OO group had larger mean values for insulin and HOMA-IR, compared to all other groups (
). There were no group differences in glucose at baseline but the OO group had larger mean values for insulin and HOMA-IR, compared to all other groups ( ).
).
Over the two-year study period, the NO group had a greater percent change in HOMA-IR (+50%) than all other groups, as shown in Table 3 ( ). The NN group had a small increase in HOMA, and the ON group had a slight decrease, while the OO group changed very little over the two-year study period. Group differences for percent change in insulin were identical to those for percent change in HOMA-IR (Table 3). When analyzing percent change in glucose, there were significant differences (
). The NN group had a small increase in HOMA, and the ON group had a slight decrease, while the OO group changed very little over the two-year study period. Group differences for percent change in insulin were identical to those for percent change in HOMA-IR (Table 3). When analyzing percent change in glucose, there were significant differences ( ) between the NN versus ON and OO groups (Table 3). The ON group was the only one to show a reduction in glucose.
) between the NN versus ON and OO groups (Table 3). The ON group was the only one to show a reduction in glucose.
The correlation between BMI change z-score and percent change in HOMA-IR was 0.49 ( ) when only the NO and ON groups were analyzed (Table 2). This relationship can be seen by comparing these groups in the scatterplot in Figure 1. Controlling for changes in V
) when only the NO and ON groups were analyzed (Table 2). This relationship can be seen by comparing these groups in the scatterplot in Figure 1. Controlling for changes in V /
/ and pubertal status did not alter the relationship significantly, as the correlation was 0.50 (Table 2). Repeating the correlations for the NO and ON groups with insulin and glucose in place of HOMA-IR revealed associations of 0.31 and 0.49, respectively.
and pubertal status did not alter the relationship significantly, as the correlation was 0.50 (Table 2). Repeating the correlations for the NO and ON groups with insulin and glucose in place of HOMA-IR revealed associations of 0.31 and 0.49, respectively.
4. Discussion
This investigation is one of the first to explore the relationship between natural progressions in weight status and changes in HOMA-IR over a two-year period in youth. Changes in weight status were not artificially manipulated by an intervention; some children started normal weight and became overweight, while some who were overweight became normal weight. When analyzing all participants as one group, there was a significant correlation ( ) between BMI change z-score and percent change in HOMA-IR even after accounting for any change in pubertal status or aerobic fitness (
) between BMI change z-score and percent change in HOMA-IR even after accounting for any change in pubertal status or aerobic fitness ( ). This suggests that changes in pubertal status and aerobic fitness did not influence the positive relationship between changes in BMI z-score and HOMA-IR. Similar associations were found in an investigation of 9-10-year-old girls followed up ten years later [24]. In that investigation, a correlation of 0.24 was reported between change in BMI and HOMA-IR and similar to the current study; these authors did not use a weight change intervention. Therefore, there appears to be a weak to moderate, positive relationship between naturally occurring alterations in weight status and IR in youth.
). This suggests that changes in pubertal status and aerobic fitness did not influence the positive relationship between changes in BMI z-score and HOMA-IR. Similar associations were found in an investigation of 9-10-year-old girls followed up ten years later [24]. In that investigation, a correlation of 0.24 was reported between change in BMI and HOMA-IR and similar to the current study; these authors did not use a weight change intervention. Therefore, there appears to be a weak to moderate, positive relationship between naturally occurring alterations in weight status and IR in youth.
4.1. Changes in HOMA-IR among the Weight Groups
As expected, HOMA-IR increased in the NO group and decreased in the ON group. The change in BMI z-score over the two-year period was remarkably similar in these groups, as it increased by 0.55 in the NO group and decreased by 0.60 in the ON group. Despite these similarities, the 7.7% reduction in HOMA-IR in the ON group was smaller than expected; this may be attributed to the relatively low HOMA-IR in the ON group at baseline. When comparing participants who became overweight (NO group) to those who remained normal weight (NN group), the 50% increase in HOMA-IR in the NO group was much larger than the 2% increase seen in the NN group. This suggests that becoming overweight is related to substantial increases in HOMA-IR that are not present when normal weight status is maintained. Taken together, these findings imply that changes in HOMA-IR may be most sensitive to natural increases in weight status, compared to decreases in or maintenance of weight status. To our knowledge, no previous investigation has directly compared participants using these weight status groups.
The lack of differences in the percent change in HOMA-IR between the OO and ON groups may be attributable to several factors. First, the mean HOMA-IR value for the ON group was 3.3 units at baseline, which was slightly lower than expected for overweight participants. This relatively low baseline value limits the capacity for further reduction in HOMA-IR in response to weight status change. Second, research has shown that insulin sensitivity declines as youth mature and pass through puberty [7, 14, 15]. In this investigation, most participants advanced by at least one pubertal stage over the two-year period. The ON group had the highest median pubertal stage at followup, which may have increased their resistance to insulin and decreased any weight-related reductions in IR. A third factor that may explain the similar changes in HOMA-IR between the OO and ON groups is the magnitude of weight status change [11, 25, 26]. For example, Reinehr et al. found significant improvements in insulin sensitivity only when obese children decreased their BMI standard deviation score by  0.5 (or 2.2 BMI kg/
0.5 (or 2.2 BMI kg/ ) over a one-year period [11]. A similar investigation found that reductions in BMI SD score of
) over a one-year period [11]. A similar investigation found that reductions in BMI SD score of  0.5 were also needed in order to elicit significant reductions in HOMA-IR after one year, and the reduction of BMI in that group was 3.2 kg/
0.5 were also needed in order to elicit significant reductions in HOMA-IR after one year, and the reduction of BMI in that group was 3.2 kg/ [26]. In the current investigation, the ON group had a mean BMI reduction of 0.9 kg/
[26]. In the current investigation, the ON group had a mean BMI reduction of 0.9 kg/ , which suggests that the weight status reduction may have been too small in magnitude to influence IR. Finally, the degree of overweight may influence the amount of change in IR. For example, Savoye et al. found reductions in HOMA-IR of 1.5 units in a group of obese participants who decreased their BMI by 1.7 kg/
, which suggests that the weight status reduction may have been too small in magnitude to influence IR. Finally, the degree of overweight may influence the amount of change in IR. For example, Savoye et al. found reductions in HOMA-IR of 1.5 units in a group of obese participants who decreased their BMI by 1.7 kg/ in one year [27]. The starting BMI for their group was 35.8 kg/
in one year [27]. The starting BMI for their group was 35.8 kg/ , however, which is much higher than the baseline BMI of 21.4 kg/
, however, which is much higher than the baseline BMI of 21.4 kg/ seen in our ON group. Taken together, these factors may explain why we saw a small percent decrease in IR in the ON group despite a 0.60-unit reduction in BMI changes z-score and subsequently, why we did not observe significant differences between the OO and ON groups.
seen in our ON group. Taken together, these factors may explain why we saw a small percent decrease in IR in the ON group despite a 0.60-unit reduction in BMI changes z-score and subsequently, why we did not observe significant differences between the OO and ON groups.
The Pearson correlation between BMI change z-score and percent change in HOMA-IR in the NO and ON participants was moderate in strength ( ). The partial correlations between these variables were nearly identical after controlling for changes in pubertal status and V
). The partial correlations between these variables were nearly identical after controlling for changes in pubertal status and V per kg of fat-free mass, suggesting that they did not influence these associations. These results disagree with cross-sectional studies that show positive relationships between pubertal status [5] and IR and negative relationships between fitness and IR in children and adolescents [16]. However, with regard to pubertal status, the changes in our participants were generally quite small which may explain their insignificant role in these relationships. The majority of participants (55%) increased by one pubertal stage over the two-year study period, while 28% did not change pubertal stage, 14% increased by two stages, and only 3 participants advanced three stages. Also, changes in fitness over the two-year period were small, ranging from −0.1 to 2.8 ml/
per kg of fat-free mass, suggesting that they did not influence these associations. These results disagree with cross-sectional studies that show positive relationships between pubertal status [5] and IR and negative relationships between fitness and IR in children and adolescents [16]. However, with regard to pubertal status, the changes in our participants were generally quite small which may explain their insignificant role in these relationships. The majority of participants (55%) increased by one pubertal stage over the two-year study period, while 28% did not change pubertal stage, 14% increased by two stages, and only 3 participants advanced three stages. Also, changes in fitness over the two-year period were small, ranging from −0.1 to 2.8 ml/ /min. This likely explains why we failed to see a relationship between changes in fitness and IR, despite a previous investigation that reported a correlation of −0.41 between V
/min. This likely explains why we failed to see a relationship between changes in fitness and IR, despite a previous investigation that reported a correlation of −0.41 between V max in mL/
max in mL/ /min and fasting insulin [16].
/min and fasting insulin [16].
4.2. HOMA-IR versus Fasting Insulin
In general, the results of this investigation were nearly identical when either percent change in HOMA-IR or percent change in insulin was used as the dependent variable in the correlation analyses (Table 2). This suggests that insulin was the driving force behind HOMA-IR, and the influence of glucose was quite small. This was also evident in the weight group comparisons at baseline, as there were no group differences in glucose but the OO group had significantly higher insulin and HOMA-IR compared to all other groups. Our findings agree with previous research showing strong associations between fasting insulin and HOMA-IR [28]. In fact, the correlation between HOMA-IR and insulin was 0.98 at baseline and 0.99 at follow-up ( ), while the correlation between HOMA-IR and glucose was 0.51 and 0.24, respectively, (
), while the correlation between HOMA-IR and glucose was 0.51 and 0.24, respectively, ( ). Thus, in our apparently healthy participants, HOMA-IR did not yield stronger associations than insulin alone, and this agrees with results from previous research supporting the use of HOMA-IR [22, 29].
). Thus, in our apparently healthy participants, HOMA-IR did not yield stronger associations than insulin alone, and this agrees with results from previous research supporting the use of HOMA-IR [22, 29].
As with any investigation, there are inherent strengths and limitations. One of the strengths of this study is the inclusion of participants who changed their weight status favorably and unfavorably as well as participants who did not change weight status. Likewise, this study is one of the few to describe the natural progression of the HOMA-IR in youth who underwent favorable changes in weight status by moving from overweight to normal weight, as the majority of research focuses on youth who move from normal weight to overweight status. A final strongpoint of this investigation is the consideration of pubertal status and aerobic fitness, as it is likely that they are related to weight status and HOMA-IR in youth.
Some limitations of this investigation include the relatively small sample sizes in each of the weight groups and the breadth of BMI values in the overweight category ( 85th percentile). If more participants were included, the overweight groups may have been divided further, into the "at risk for overweight" and "overweight" groups, as described by the CDC [23]. Another limitation is the use of self-report measures of pubertal status. While some research has reported high correlations (
85th percentile). If more participants were included, the overweight groups may have been divided further, into the "at risk for overweight" and "overweight" groups, as described by the CDC [23]. Another limitation is the use of self-report measures of pubertal status. While some research has reported high correlations ( ) among self- and physician-generated ratings of Tanner stage in adolescent girls [30], other studies have weaker relationships [31]. Also, there were slight inequities for age and race, which may have impacted HOMA-IR values in this investigation, as research shows that black females had higher insulin and HOMA-IR values compared to White girls, regardless of pubertal stage [24]. Additionally, cut-points ranging from 3.16 to 4.0 for HOMA-IR have been used to classify IR in children and adolescents, yet there is no universally accepted value for use in youth [2, 22]. This complicates the interpretation of results, as different conclusions can be formulated depending upon which cut-point is used. As a result, we did not dichotomize individuals as insulin resistant or not. Once a consensus is reached regarding these cut-points, normal and overweight participants could be compared using these insulin resistance classifications. The weight group analysis could be considered both a strength, as mentioned above, or a weakness. As a weakness, the groups have limited power, which could skew our findings. Also, although determined a priori, the criteria for allocation to a group were artificial. There is no reason why a person in the normal group cannot have a large change in BMI; yet because they do not cross the cut-point, they do not enter the "overweight" group. Thus our grouping does not allow for any conclusions regarding a large change in BMI, without a change in categorization. Finally, we are unable to determine temporality of these relationships using the current design. As a result, we cannot determine if changes in weight status caused changes in HOMA-IR or vice versa and instead can only comment upon their associations.
) among self- and physician-generated ratings of Tanner stage in adolescent girls [30], other studies have weaker relationships [31]. Also, there were slight inequities for age and race, which may have impacted HOMA-IR values in this investigation, as research shows that black females had higher insulin and HOMA-IR values compared to White girls, regardless of pubertal stage [24]. Additionally, cut-points ranging from 3.16 to 4.0 for HOMA-IR have been used to classify IR in children and adolescents, yet there is no universally accepted value for use in youth [2, 22]. This complicates the interpretation of results, as different conclusions can be formulated depending upon which cut-point is used. As a result, we did not dichotomize individuals as insulin resistant or not. Once a consensus is reached regarding these cut-points, normal and overweight participants could be compared using these insulin resistance classifications. The weight group analysis could be considered both a strength, as mentioned above, or a weakness. As a weakness, the groups have limited power, which could skew our findings. Also, although determined a priori, the criteria for allocation to a group were artificial. There is no reason why a person in the normal group cannot have a large change in BMI; yet because they do not cross the cut-point, they do not enter the "overweight" group. Thus our grouping does not allow for any conclusions regarding a large change in BMI, without a change in categorization. Finally, we are unable to determine temporality of these relationships using the current design. As a result, we cannot determine if changes in weight status caused changes in HOMA-IR or vice versa and instead can only comment upon their associations.
5. Conclusion
In youth, changes in BMI z-score and percent change in HOMA-IR over two years are positively related. Specifically, participants who underwent natural increases in weight status and became overweight had substantial increases in HOMA-IR, and the magnitude of these changes was greater than those for youth who decreased or maintained their weight status. These results reinforce the importance of preventing overweight during childhood.
References
Druet C, Dabbas M, Baltakse V: Insulin resistance and the metabolic syndrome in obese French children. Clinical Endocrinology. 2006, 64 (6): 672-678. 10.1111/j.1365-2265.2006.02526.x.
Lee JM, Okumura MJ, Davis MM, Herman WH, Gurney JG: Prevalence and determinants of insulin resistance among U.S. adolescents: a population-based study. Diabetes Care. 2006, 29 (11): 2427-2432. 10.2337/dc06-0709.
Bacha F, Saad R, Gungor N, Arslanian SA: Are obesity-related metabolic risk factors modulated by the degree of insulin resistance in adolescents?. Diabetes Care. 2006, 29 (7): 1599-1604. 10.2337/dc06-0581.
Gower BA, Nagy TR, Goran MI: Visceral fat, insulin sensitivity, and lipids in prepubertal children. Diabetes. 1999, 48 (8): 1515-1521. 10.2337/diabetes.48.8.1515.
Roemmich JN, Clark PA, Lusk M: Pubertal alterations in growth and body composition. VI. Pubertal insulin resistance: relation to adiposity, body fat distribution and hormone release. International Journal of Obesity. 2002, 26 (5): 701-709. 10.1038/sj.ijo.0801975.
Thorsdottir I, Gunnarsdottir I, Palsson GI, Johannsson E: Anthropometric predictors of serum fasting insulin in 9- and 15-year-old children and adolescents. Nutrition, Metabolism and Cardiovascular Diseases. 2006, 16 (4): 263-271. 10.1016/j.numecd.2005.09.003.
Travers SH, Jeffers BW, Bloch CA, Hill JO, Eckel RH: Gender and Tanner stage differences in body composition and insulin sensitivity in early pubertal children. The Journal of Clinical Endocrinology & Metabolism. 1995, 80 (1): 172-178. 10.1210/jc.80.1.172.
Young-Hyman D, Schlundt DG, Herman L, De Luca F, Counts D: Evaluation of the insulin resistance syndrome in 5- to 10-year-old overweight/obese African-American children. Diabetes Care. 2001, 24 (8): 1359-1364. 10.2337/diacare.24.8.1359.
Johnson MS, Figueroa-Colon R, Huang TT-K, Dwyer JH, Goran MI: Longitudinal changes in body fat in African American and Caucasian children: influence of fasting insulin and insulin sensitivity. The Journal of Clinical Endocrinology & Metabolism. 2001, 86 (7): 3182-3187. 10.1210/jc.86.7.3182.
Lazzer S, Vermorel M, Montaurier C, Meyer M, Boirie Y: Changes in adipocyte hormones and lipid oxidation associated with weight loss and regain in severely obese adolescents. International Journal of Obesity. 2005, 29 (10): 1184-1191. 10.1038/sj.ijo.0802977.
Reinehr T, Kiess W, Kapellen T, Andler W: Insulin sensitivity among obese children and adolescents, according to degree of weight loss. Pediatrics. 2004, 114 (6): 1569-1573. 10.1542/peds.2003-0649-F.
National Health Examination Survey : Vital and Health Statistics. Series 11. 1974, N. D. o. H., Education, and Welfare (DHEW), Publication #74-1614:2-3
Slaughter MH, Lohman TG, Boileau RA: Skinfold equations for estimations of body fatness in children and youth. Human Biology. 1988, 60 (5): 709-723.
Cook JS, Hoffman RP, Stene MA, Hansen JR: Effects of maturational stage on insulin sensitivity during puberty. The Journal of Clinical Endocrinology & Metabolism. 1993, 77 (3): 725-730. 10.1210/jc.77.3.725.
Guzzaloni G, Grugni G, Mazzilli G, Moro D, Morabito F: Comparison between
 -cell function and insulin resistance indexes in prepubertal and pubertal obese children. Metabolism. 2002, 51 (8): 1011-1016. 10.1053/meta.2002.34029.
-cell function and insulin resistance indexes in prepubertal and pubertal obese children. Metabolism. 2002, 51 (8): 1011-1016. 10.1053/meta.2002.34029.Allen DB, Nemeth BA, Clark RR, Peterson SE, Eickhoff J, Carrel AL: Fitness is a stronger predictor of fasting insulin levels than fatness in overweight male middle-school children. Journal of Pediatrics. 2007, 150 (4): 383-387. 10.1016/j.jpeds.2006.12.051.
Petersen AC, Crockett L, Richards M, Boxer A: A self-report measure of pubertal status: reliability, validity, and initial norms. Journal of Youth and Adolescence. 1988, 17 (2): 117-133. 10.1007/BF01537962.
McMurray RG, Guion WK, Ainsworth BE, Harrell JS: Predicting aerobic power in children. A comparison of two methods. Journal of Sports Medicine and Physical Fitness. 1998, 38 (3): 227-233.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC: Homeostasis model assessment: insulin resistance and
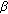 -cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985, 28 (7): 412-419. 10.1007/BF00280883.
-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985, 28 (7): 412-419. 10.1007/BF00280883.Atabek ME, Pirgon O: Assessment of insulin sensitivity from measurements in fasting state and during an oral glucose tolerance test in obese children. Journal of Pediatric Endocrinology and Metabolism. 2007, 20 (2): 187-195. 10.1515/JPEM.2007.20.2.187.
Gungor N, Saad R, Janosky J, Arslanian S: Validation of surrogate estimates of insulin sensitivity and insulin secretion in children and adolescents. Journal of Pediatrics. 2004, 144 (1): 47-55. 10.1016/j.jpeds.2003.09.045.
Keskin M, Kurtoglu S, Kendirci M, Atabek ME, Yazici C: Homeostasis model assessment is more reliable than the fasting glucose/insulin ratio and quantitative insulin sensitivity check index for assessing insulin resistance among obese children and adolescents. Pediatrics. 2005, 115 (4): e500-e503. 10.1542/peds.2004-1921.
Centers for Disease Control and Prevention , National Center for Health Statistics : April 2007, http://www.cdc.gov/GROWTHCHARTS/
Klein DJ, Friedman LA, Harlan WR: Obesity and the development of insulin resistance and impaired fasting glucose in black and white adolescent girls: a longitudinal study. Diabetes Care. 2004, 27 (2): 378-383. 10.2337/diacare.27.2.378.
Reinehr T, Andler W: Cortisol and its relation to insulin resistance before and after weight loss in obese children. Hormone Research. 2004, 62 (3): 107-112. 10.1159/000079841.
Reinehr T, Andler W: Changes in the atherogenic risk factor profile according to degree of weight loss. Archives of Disease in Childhood. 2004, 89 (5): 419-422. 10.1136/adc.2003.028803.
Savoye M, Shaw M, Dziura J: Effects of a weight management program on body composition and metabolic parameters in overweight children: a randomized controlled trial. Journal of the American Medical Association. 2007, 297 (24): 2697-2704. 10.1001/jama.297.24.2697.
Wilson DM, Wang Y, Cullen KW: Assessing weight-related biochemical cardiovascular risk factors in African-American girls. Obesity Research. 2004, 12 (S9): 73S-83S. 10.1038/oby.2004.271.
McAuley KA, Mann JI, Chase JG: Point: HOMA—satisfactory for the time being: HOMA: the best bet for the simple determination of insulin sensitivity, until something better comes along. Diabetes Care. 2007, 30 (9): 2411-2413. 10.2337/dc07-1067.
Brooks-Gunn J, Warren MP, Rosso J, Gargiulo J: Validity of self-report measures of girls' pubertal status. Child Development. 1987, 58 (3): 829-841. 10.2307/1130220.
Desmangles J-C, Lappe JM, Lipaczewski G, Haynatzki G: Accuracy of pubertal tanner staging self-reporting. Journal of Pediatric Endocrinology and Metabolism. 2006, 19 (3): 213-221. 10.1515/JPEM.2006.19.3.213.
Acknowledgment
Data collection was supported by Grant no. NR01-1837 from NIH/NINR.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is published under license to BioMed Central Ltd. This is an Open Access article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Ondrak, K.S., McMurray, R.G., Battaglini, C.L. et al. The Relationship between Changes in Weight Status and Insulin Resistance in Youth. Int J Pediatr Endocrinol 2009, 862061 (2009). https://doi.org/10.1155/2009/862061
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1155/2009/862061

 -cell function and insulin resistance indexes in prepubertal and pubertal obese children. Metabolism. 2002, 51 (8): 1011-1016. 10.1053/meta.2002.34029.
-cell function and insulin resistance indexes in prepubertal and pubertal obese children. Metabolism. 2002, 51 (8): 1011-1016. 10.1053/meta.2002.34029.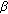 -cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985, 28 (7): 412-419. 10.1007/BF00280883.
-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985, 28 (7): 412-419. 10.1007/BF00280883.